New Zealanders are being promised early access to clinical trials for a ground-breaking new method of destroying cancer.
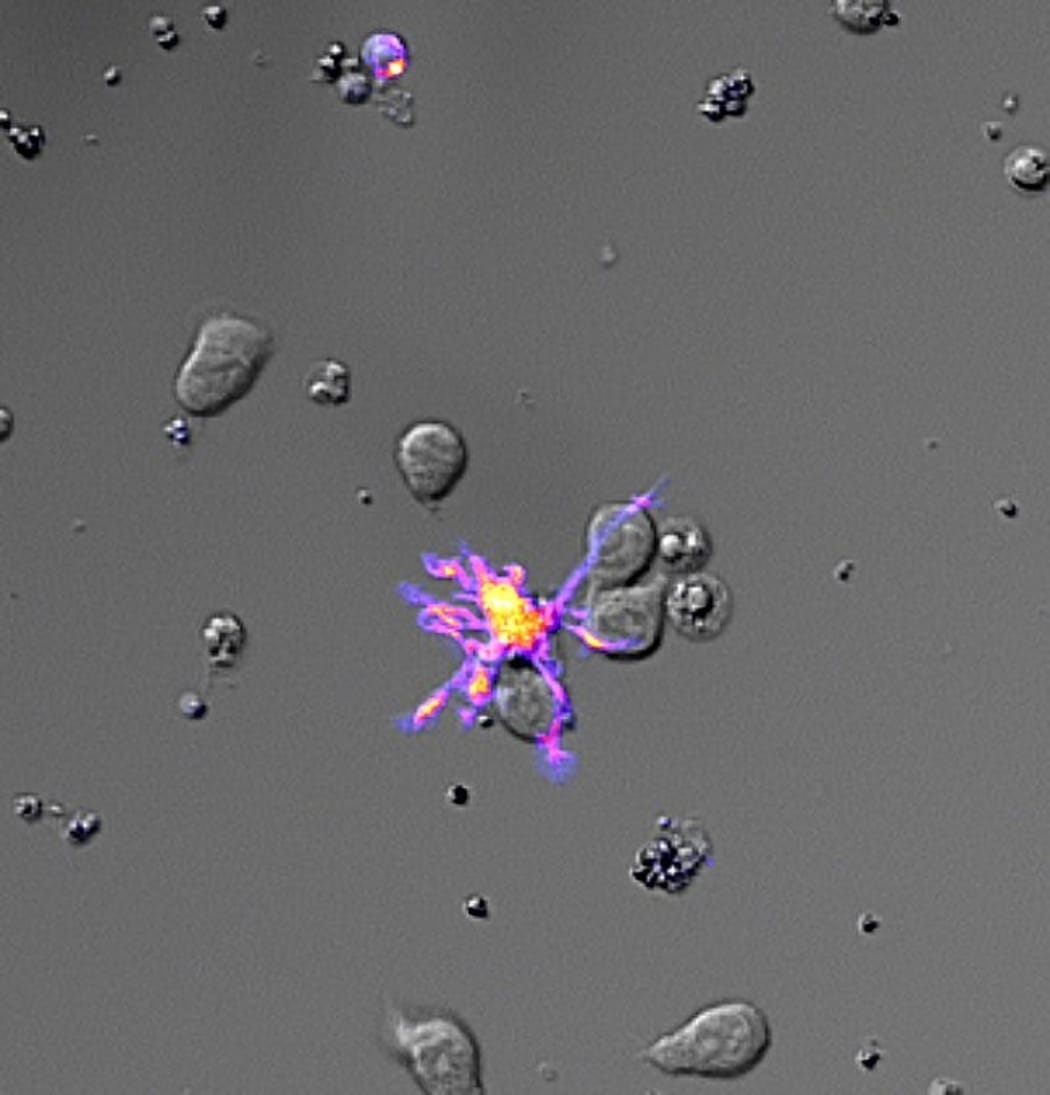
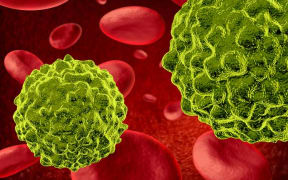
A microscopy photo shows a dendritic cell prompting T-cells to form a large population to fight a disease. Photo: Malaghan Institute
It will be done under a New Zealand-Chinese biotech joint venture unveiled this afternoon.
Under the deal, Wellington-based Malaghan Institute joins China's Hunan Zhaotai Medical Group in taking one of the latest research techniques for fighting cancer to trial both here and elsewhere as a potential new way of treating cancer.
That technique is immunotherapy, an approach that is sweeping the world with drugs like Keytruda and Opdivo, which were funded in New Zealand last year.
But, whereas they are compounds given to patients, another new breakthrough area involves taking a person's own immune cells, modifying them and then injecting them back into the patient where they target and kill cancer.

Prof Graham Le Gros Photo: Supplied
Malaghan director Graham Le Gros said it was exciting.
"The patient's own cells, immune cells, which don't seem to be able to see the cancer so well, they're just taken out and they can be genetically modified, through some genetic tricks, actually to see the cancer, or a certain B-cell form of the cancer, and then they are then grown up and put back in, and away they go and get rid of the cancer."
He said it was not done here but had been developed by Hunan Zhaotai in China, where it was already keeping some patients alive.
"They've already taken them through the preliminary phase-one trials. So they've been in patients, shown that they can work in patients in China, so a lot of issues have been resolved.
"What they need now is support in taking it to the next level for international distribution and for getting up to a level where it can be used clinically anywhere in the world."
Professor Le Gros said his institute's role would be to ensure the therapy met international regulatory standards and could be used safely.
He said in New Zealand it would mean patients with leukaemias and some other cancers would gain early access to clinical trials, and hence treatment.
"We anticipate that this will lead to access for New Zealand patients for clinical trials here in New Zealand."
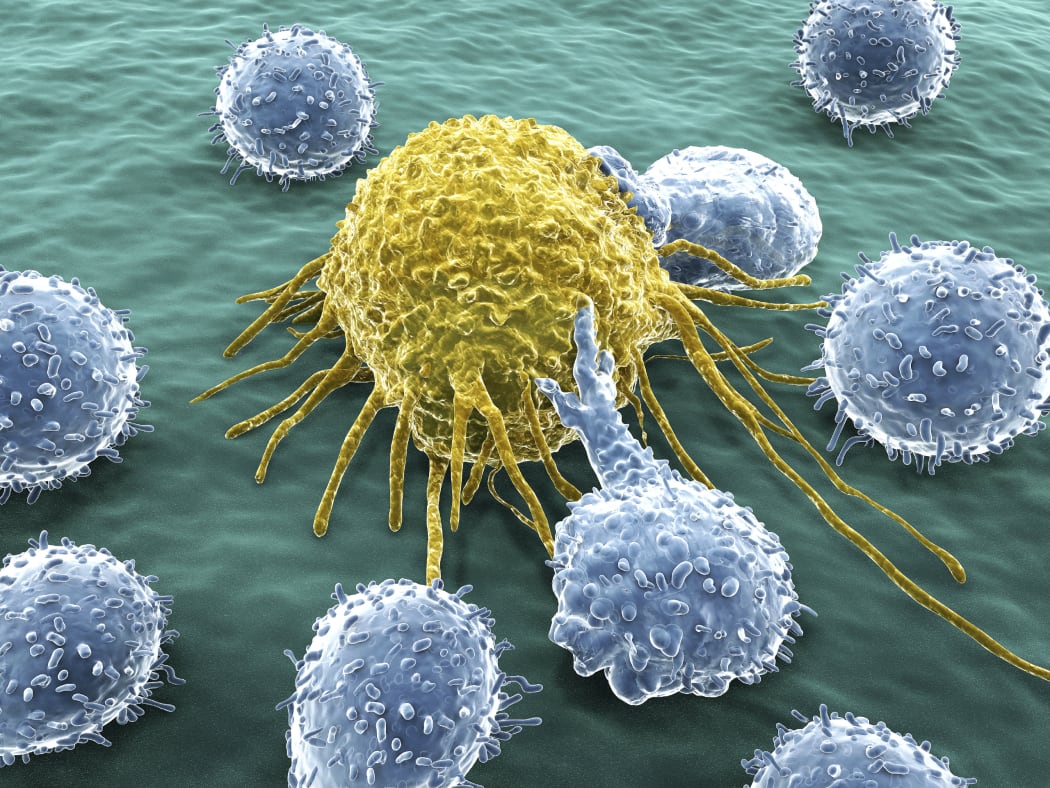
A T-cell latching onto a yellow cancer cell to kill it Photo: Malaghan Institute
But Prof Le Gros said it was not possible to give a timeframe.
"I can't say when, not because it's not going to happen, just because it just raises expectations and you've got to realise there's a lot of people out there who are suffering from various forms of cancer. They want to know well where, what, how, and we just can't say ethically. It's just not appropriate for us to say anything at this point."
Health Research Council chief executive Kath McPherson agreed it would bring major opportunities New Zealand's way.
"We will get the first access to those drugs and trial before things are spread to the rest of the world so it's a really unique opportunity."
Cancer Society medical director Chris Jackson stressed these were research therapies primarily, and research was the reason they were expected to become available in this country.
That aside, he said it was a great opportunity for New Zealand to be at the cutting edge of immune therapy research.
"While they have shown a lot of promise in diseases like pancreas cancer, mesothelioma and indeed leukaemias, that we're not yet certain what the clinical trials on offer will be, but it will give New Zealand patients and clinicians an opportunity to develop new research therapies in this area."
Dr Jackson said it should lead to a new wave of clinical research in immunotherapy in this country.
"One of the key things is going to be ensuring that the access to this treatment is rolled out around the country and that patients will be able to access these clinical trials no matter where they are in the country."
He added more funding would be needed to boost capacity for clinical trials.